Abstract
The liability architecture for vaccine manufacturers in the United States is dominated by two statutory regimes: the Public Readiness and Emergency Preparedness (PREP) Act and the National Childhood Vaccine Injury Act (NCVIA), which established the Vaccine Injury Compensation Program (VICP). These frameworks grant sweeping immunity to manufacturers, channeling claims into specialized compensation systems. Yet both contain pathways—executive, administrative, and judicial—that can prospectively or retrospectively strip immunity.
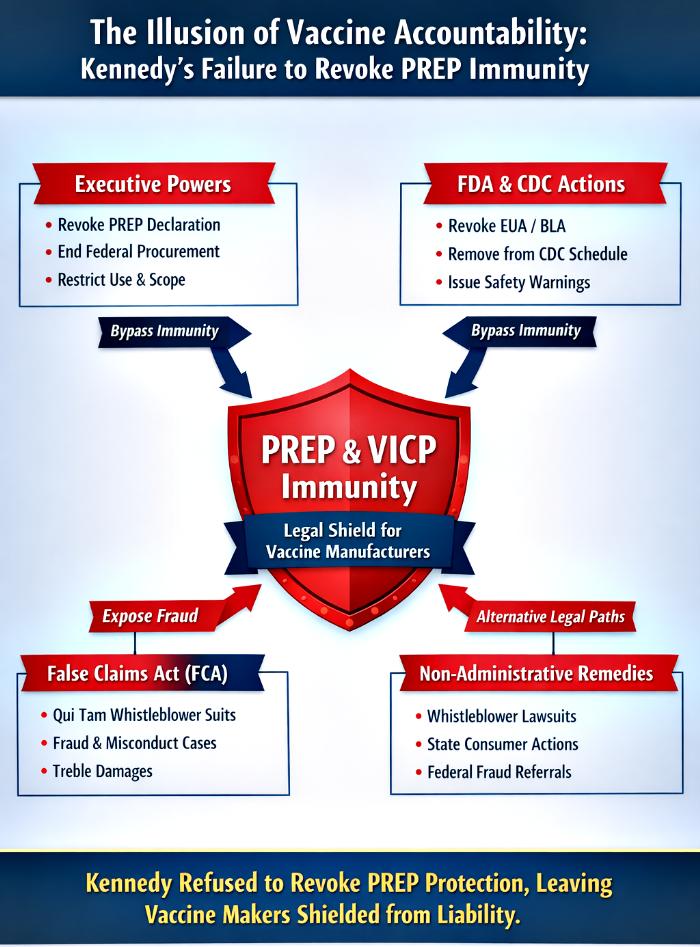
This article examines the full spectrum of powers available to Secretary Kennedy under the separation of powers doctrine: executive authority through PREP declarations, administrative oversight via FDA and CDC, quasi‑judicial enforcement through penalties and investigations, and parallel remedies such as the False Claims Act (FCA). It demonstrates that Kennedy possessed direct and effective tools to revoke immunity but instead chose a procedurally flawed ACIP maneuver that predictably collapsed under judicial review in AAP v Kennedy. He is still following the same script without actually doing anything to safeguard the interests of US citizens.
By mapping these pathways and analyzing their implications, the article argues that Kennedy’s refusal to exercise his strongest executive lever—the revocation of the PREP Declaration—represents a profound failure of governance. The result is continued protection for vaccine manufacturers, leaving citizens reliant on weaker remedies such as FCA suits, whistleblower actions, and state consumer claims.
Introduction
Constitutional governance rests on the separation of powers: legislatures make laws, executives enforce them, and judiciaries interpret them. Within this framework, vaccine liability regimes in the United States balance two imperatives: incentivizing rapid vaccine development and ensuring accountability for misconduct. The PREP Act grants broad immunity to manufacturers of designated countermeasures, while the NCVIA channels injury claims into the VICP.
Yet immunity is not absolute. The Secretary of Health and Human Services (HHS) can revoke or amend PREP declarations, FDA can withdraw authorizations, CDC can alter immunization schedules, quasi‑judicial enforcement can expose misconduct, and the False Claims Act provides a parallel fraud‑based enforcement channel. These mechanisms bypass statutory channeling and open the door to traditional litigation.
Despite possessing these tools, Secretary Kennedy chose not to revoke the PREP Declaration for COVID vaccines—the most direct and effective pathway to strip immunity. Instead, he pursued a controversial ACIP maneuver that lacked procedural legitimacy and was stayed in AAP v Kennedy. This article dissects Kennedy’s available powers, his actual actions, and the consequences of his failure.
Remedies Through Executive, Administrative, And Quasi‑Judicial Powers
Table 1 — EAQ Powers Of Secretary Kennedy
| Executive Powers | Administrative Powers | Quasi‑Judicial Powers |
|---|---|---|
| Issue or revoke PREP Act declarations (direct authority to grant/remove immunity). | FDA withdrawal of EUA/BLA; CDC/ACIP schedule changes. | Impose fines, penalties, sanctions; launch investigations into fraud or concealment. |
| Narrow scope of PREP declaration (limit persons, uses, geography). | HRSA programmatic delisting or prioritization changes in VICP intake. | Refer cases for criminal prosecution; support whistleblower suits. |
| Stop federal procurement or remove vaccines from federal programs. | Issue safety advisories, enforcement letters, warning notices. | Provide evidentiary records for state consumer protection actions. |
| Coordinate multi‑agency sequence (PREP revocation + FDA withdrawal + CDC delisting + procurement stop). | Adjust programmatic intake and prioritization. | Enable retrospective liability through misconduct findings. |
Discussion:
Kennedy’s executive authority under the PREP Act was the most direct lever—a simple revocation of the COVID vaccine declaration would have stripped immunity instantly. Administrative tools such as FDA withdrawal or CDC schedule changes were secondary but still potent. Quasi‑judicial enforcement could expose fraud and misconduct, feeding evidence into litigation. Together, these powers formed a comprehensive arsenal to dismantle vaccine immunity.
Analysis:
(1) Executive Primacy: The PREP Declaration functions as an on/off switch for immunity. Kennedy’s refusal to revoke it represents a conscious abdication of responsibility.
(2) Administrative Support: FDA and CDC actions could have narrowed immunity, but Kennedy relied on ACIP theatrics that collapsed under judicial scrutiny.
(3) Quasi‑Judicial Enforcement: Investigations and penalties remain underutilized, leaving misconduct unpunished and immunity intact.
FDA And CDC Administrative Pathways
Table 2 — FDA And CDC Remedies
| FDA Actions | CDC/ACIP Actions | Procedural Requirements |
|---|---|---|
| Withdraw or revoke EUA/BLA for vaccines. | ACIP votes to remove vaccine from immunization schedule; CDC adopts recommendation. | Must follow proper administrative procedure: notice, comment, scientific review. |
| Narrow indications or relabel vaccines. | Issue CDC guidance narrowing schedule inclusion. | Failure to follow procedure leads to judicial stay (AAP v Kennedy). |
| Issue enforcement letters, warning notices, or safety advisories. | Adjust HRSA/CDC programmatic intake prioritization for VICP claims. | Courts intervene if due process or statutory mandates are violated. |
Discussion:
FDA and CDC wield indirect but significant powers. Withdrawal of authorizations or removal from immunization schedules reshapes the liability landscape by narrowing contexts where immunity applies. However, these actions require strict adherence to administrative procedure. Kennedy’s reliance on ACIP votes without procedural compliance guaranteed judicial invalidation.
Analysis:
(1) FDA Leverage: Withdrawal of EUA/BLA would have removed COVID vaccines from authorized use, exposing manufacturers to liability. Kennedy did not pursue this.
(2) CDC Fragility: ACIP votes are procedurally vulnerable. Kennedy’s reliance on this mechanism was a calculated but hollow gesture.
(3) Judicial Oversight: Courts swiftly stayed Kennedy’s ACIP maneuver, underscoring the futility of bypassing procedural safeguards.
False Claims Act: A Parallel Enforcement Channel
Table 3 — False Claims Act Remedies
| Mechanism | Actor | Effect on Immunity | Evidence Needed | Practical Consequence |
|---|---|---|---|---|
| FCA civil action (qui tam or DOJ suit) | DOJ, whistleblowers | Does not directly revoke PREP/VICP immunity but exposes fraud outside those regimes | False billing records, misrepresentation of safety/efficacy, concealment of adverse data | Treble damages, civil penalties, evidentiary record supporting fraud/misconduct claims |
| Whistleblower disclosures under FCA | Private relators | Creates independent litigation channel | Internal communications, suppressed adverse event data, falsified submissions | Strengthens plaintiffs’ ability to argue fraud defeats statutory channeling |
| DOJ enforcement | Federal prosecutors | Parallel enforcement outside PREP/VICP | Criminal referrals, administrative findings | Adds weight to judicial findings that can strip immunity retrospectively |
Discussion:
The FCA bypasses PREP and VICP entirely because it targets fraud against the government, not personal injury claims. Successful FCA actions produce robust evidence of fraud—internal emails, falsified submissions, suppressed adverse data—that courts can use to retrospectively defeat PREP or VICP immunity.
Analysis:
(1) Independent Enforcement Channel: FCA suits operate outside statutory channeling, making them one of the few remedies immune to PREP/VICP barriers.
(2) Evidentiary Bridge: FCA findings create the factual predicate for courts to pierce immunity in related injury cases.
(3) Instrumental In Parallel Tort Claims The FCA enables tort liability by creating a statutory mechanism that both proves and channels civil wrongdoing—its elements (a false representation, materiality, presentment to the government, and scienter) mirror common-law fraud elements so a successful FCA case often supplies the core factual and evidentiary predicates for parallel tort claims (fraud, conspiracy, unjust enrichment); additionally, qui tam proceedings unlock expansive pre-trial discovery and can produce judicial findings on falsity and intent that support collateral tort actions or equitable remedies, while state-law claims can be asserted alongside or where federal coverage is incomplete to capture private harms and restitution beyond the FCA’s statutory remedies.
Non‑Administrative Remedies: The Last Resort
Citizens now face reliance on weaker remedies outside the executive and administrative sphere:
(1) Whistleblower suits exposing concealed safety data.
(2) State consumer protection actions challenging deceptive practices.
(3) Federal enforcement referrals generating evidentiary records for litigation.
These remedies are reactive, slow, and dependent on judicial findings of fraud or misconduct. They lack the immediacy of executive revocation and rarely succeed in stripping immunity.
Conclusion
Secretary Kennedy possessed the most powerful tool to dismantle vaccine manufacturer immunity: revocation of the PREP Declaration for COVID-19 shots. This action required no legislative approval and would have instantly exposed manufacturers to traditional tort liability. Instead, Kennedy staged a symbolic ACIP maneuver that predictably collapsed under judicial review.
By refusing to exercise his executive authority, Kennedy has failed U.S. citizens and betrayed his core voters. His administration remains aligned with the vaccine lobby, preserving immunity and shielding manufacturers from accountability. The result is a liability regime where citizens must rely on weak remedies—FCA suits, whistleblower actions, and state consumer claims—while the pharmaceutical cartel continues to dominate.
Kennedy’s failure is not procedural—it is political. He chose drama over substance, optics over accountability, and lobby interests over public trust. The consequence is clear: vaccine immunity remains intact, and the promise of accountability has been sacrificed. The verdict of history will be unforgiving.
In these dark times of medical tyranny, systemic gaslighting, and the denial of remedies to the vaccine-injured, the HVBI Framework emerges as a guiding light. The path forward is clear—let the HVBI Framework illuminate the way.
