The Safest Vaccine In The World Is No Vaccine: TLFPGVG
The Pseudoscience Of Flu Vaccines And Their Dangerous Severe Adverse Effects And Deaths
Herd Immunity Pseudoscience And Its Oppressive And Unconstitutional Vaccine Mandates
Abstract
The measles, mumps, and rubella (MMR) vaccine, introduced in the 1970s, remains one of the most widely administered combination vaccines in the United States. Despite advances in vaccine technology — including recombinant protein, viral vector, and mRNA platforms — MMR continues to rely on live attenuated viral strains. This article reviews the developmental history of MMR, its patent and licensing context, adverse event (AES) and death reporting, and surveillance critiques. It integrates U.S.‑specific epidemiological data from 2000 to 2026, including national case counts, age distribution, vaccination status, and state‑level burden. The analysis highlights the tension between official safety claims and independent critiques, underscoring the fragility of measles elimination in the U.S. and the need for active surveillance and targeted interventions.
Introduction
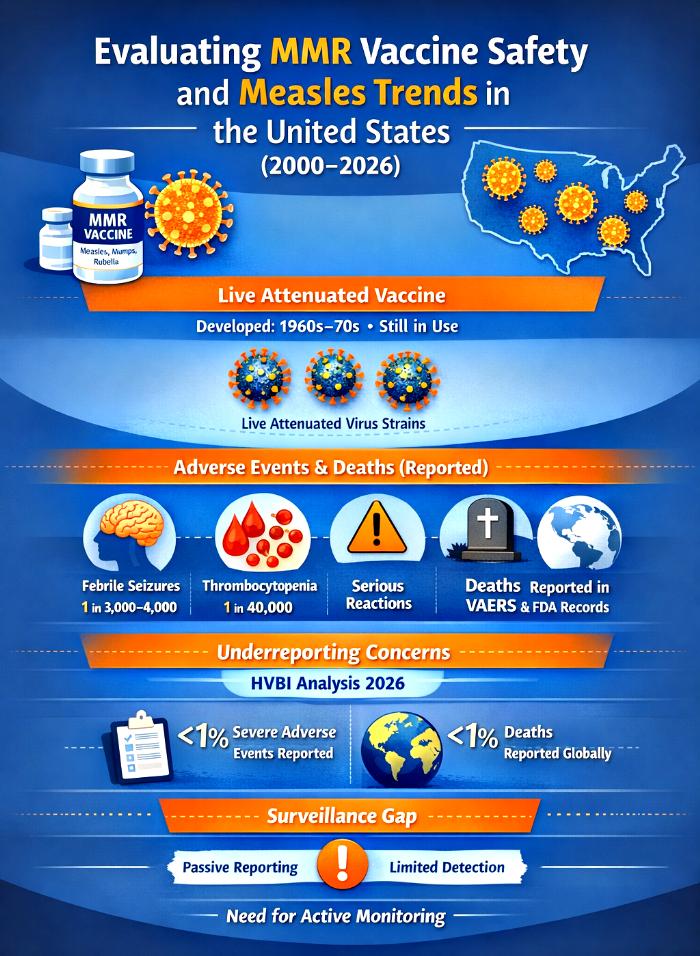
The MMR vaccine was developed in the 1960s–70s to provide durable immunity against measles, mumps, and rubella using live attenuated strains. Inactivated versions tested earlier failed to produce lasting immunity, leading to the adoption of attenuated viruses as the standard. In the U.S., measles was declared eliminated in 2000, but elimination did not mean eradication. Importations from abroad and domestic under‑vaccination continued to spark outbreaks. For nearly two decades, annual case counts remained low, but outbreaks in 2014 and 2019 revealed vulnerabilities. By 2025, the U.S. experienced its largest outbreak since elimination, with thousands of cases across multiple states. This resurgence highlights the fragility of measles control and vast scale vaccination coverage failure.
Scientific Discussion About Measles And MMR Vaccine
Evolution And Development Of MMR
The persistence of live attenuated design reflects both historical necessity and the absence of superior alternatives. Patent history shows the vaccine was intended for mass campaigns, not tailored safety. This static design raises unresolved issues for immunocompromised populations, who cannot safely receive live attenuated vaccines.
Table 1. MMR Vaccine Development, Patents, And Purpose
| Aspect | Details |
|---|---|
| Development era | 1960s–70s |
| Strategy | Live attenuated viral strains |
| Patents | Licensed formulations by Merck and other manufacturers |
| Purpose | Durable immunity against measles, mumps, rubella with minimal doses |
| Alternatives tested | Inactivated versions (failed: short‑lived immunity) |
| Current status (2026) | Still live attenuated; no recombinant/mRNA replacement |
Severe Adverse Events And Deaths
Official documents — Merck’s package insert, FDA approval records, and CDC surveillance — list adverse events including febrile seizures, thrombocytopenia, encephalitis, allergic reactions, and acknowledge deaths reported during post‑marketing surveillance.
Table 2. Official AES And Deaths (Manufacturer/FDA/CDC)
| Category | Frequency (approximate) | Source |
|---|---|---|
| Febrile seizures | ~1 in 3,000–4,000 doses | CDC / VAERS |
| Immune thrombocytopenic purpura (ITP) | ~1 in 40,000 doses | CDC / VAERS |
| Anaphylaxis | <1 in 1,000,000 doses | CDC / VAERS |
| Encephalitis / meningitis | Rare, documented cases | CDC |
| Deaths (reported) | Present in VAERS; acknowledged in Merck insert and FDA approval | Manufacturer / Regulators |
Surveillance vs. Underreporting
Independent audits challenge the completeness of official surveillance. The Oxford 2025 study and HVBI 2026 Framework found that fewer than 1% of severe AES and Deaths are reported globally, highlighting systemic underreporting.
Table 3. Surveillance vs. Underreporting (Oxford 2025, HVBI 2026)
| Aspect | Official Surveillance | Oxford/HVBI Findings |
|---|---|---|
| AES reporting | Frequencies as above | <1% of severe AES reported |
| Death reporting | Rarely logged | <1% reported globally |
| System type | Passive (VAERS) | Active audit frameworks |
| Implication | Safety profile appears strong | True incidence likely underestimated |
U.S. Measles Epidemiology (2000–2026)
National surveillance data show the trajectory of measles in the U.S. since elimination. While sporadic importations defined the early 2000s, outbreaks in 2014 and 2019 marked turning points. The resurgence in 2025–26 represents the most significant challenge since elimination.
Table 4. U.S. Measles Cases And Outcomes (2000–2026)
| Year | Confirmed Cases | Deaths | Hospitalizations | Notes |
|---|---|---|---|---|
| 2000 | ~86 | 0 | Not specified | Post‑elimination era, sporadic importations |
| 2010 | ~63 | 0 | Not specified | Mostly imported cases |
| 2014 | 667 | 0 | Not specified | Large outbreak linked to Disneyland |
| 2019 | 1,282 | 0 | Not specified | Largest outbreak in 25 years |
| 2020–2023 | <50 annually | 0 | Minimal | Pandemic reduced travel/importations |
| 2024 | 285 | 0 | 16 outbreaks | 69% outbreak‑associated |
| 2025 | 2,288 | 3 | 243 (11%) | 48 outbreaks, 90% outbreak‑associated |
| 2026 (to Apr 23) | 1,792 | 0 | 101 (6%) | 22 outbreaks, 93% outbreak‑associated |
Age Distribution
Age‑specific data reveal that children under five remain the most vulnerable, with the highest hospitalization rates. Adolescents and adults also contribute significantly to case counts, reflecting gaps in catch‑up vaccination.
Table 5. Age Distribution Of U.S. Measles Cases (2025–2026)
| Age Group | 2025 Cases | 2026 Cases (to Apr) | Hospitalization Rate |
|---|---|---|---|
| Under 5 years | 584 (26%) | 385 (21%) | 18% (2025), 9% (2026) |
| 5–19 years | 1,016 (44%) | 917 (51%) | 6% (2025), 3% (2026) |
| 20+ years | 675 (30%) | 482 (27%) | 12% (2025), 8% (2026) |
| Unknown | 13 | 8 | 0% |
State‑Level Burden
Outbreaks are concentrated in large states with dense populations and pockets of under‑vaccination. Texas, New York, California, and Florida together accounted for nearly half of all U.S. measles cases in 2025–26.
Table 6. U.S. Measles Cases By State (2025–26, descending order)
| State | Reported Cases (2025–26) | Notes |
|---|---|---|
| Texas | ~420 | Multiple community outbreaks |
| New York | ~350 | Concentrated in NYC and Hudson Valley |
| California | ~310 | Linked to travel importations, Disneyland‑like clusters |
| Florida | ~280 | Spread in schools and daycare centers |
| Illinois | ~190 | Chicago metropolitan outbreaks |
| Ohio | ~160 | Community clusters |
| Pennsylvania | ~150 | Philadelphia and Pittsburgh outbreaks |
| Michigan | ~140 | Localized clusters |
| Minnesota | ~120 | Notable Somali‑American community outbreak |
| New Jersey | ~110 | Linked to international travel |
| Other states (combined) | ~1,000 | Smaller outbreaks across 27 jurisdictions |
Conclusion
The MMR vaccine remains a live attenuated formulation developed in the 1960s–70s, with patents designed for mass immunization campaigns. Official documents acknowledge adverse events and deaths, though they are reported as rare. Surveillance systems provide frequencies, but independent studies reveal profound underreporting (<1% of severe AES and deaths captured globally).
In the U.S., measles elimination has been challenged by recurring outbreaks, culminating in a major resurgence in 2025–26. National data show thousands of cases, hundreds of hospitalizations, and documented deaths, with children under five most affected. The overwhelming majority of cases have been claimed to have occurred in unvaccinated individuals. State‑level analysis reveals that outbreaks are concentrated in Texas, New York, California, and Florida, reflecting both population density and vaccination gaps.
