The Safest Vaccine In The World Is No Vaccine: TLFPGVG
Beyond The Banner Of Measles: Why Bangladesh’s Measles‑Like Symptoms (MLS) Crisis Is Not Measles
Abstract
Bangladesh’s biosafety landscape is characterized by a paradox: the country has invested heavily in BSL-3 laboratories, supported by international collaborations, yet it lacks any operational BSL-4 facilities. This absence has profound implications for national and regional biosecurity, particularly given Bangladesh’s endemic exposure to high-risk pathogens such as Nipah virus and Avian Influenza H5N1. This article explores the historical trajectory of Bangladesh’s BSL-3 infrastructure, its reliance on global partnerships, and the gradual shift toward independent management. It also examines vaccine-related collaborations with China, situating them within broader geopolitical dynamics of biosafety and biosecurity. Through a detailed analysis of key laboratory facilities and partnerships, the article argues that Bangladesh’s biosafety system is both strengthened and constrained by its dependence on external actors. The conclusion highlights the urgent need for sovereign capacity-building to ensure compliance with international standards while safeguarding against potential misuse of gain-of-function research.
Introduction
The global biosafety architecture is unevenly distributed, with advanced containment facilities concentrated in a handful of countries. Bangladesh, despite being a hotspot for zoonotic diseases, remains without a BSL-4 laboratory. Instead, the country has developed a network of BSL-3 laboratories, many of which were established through international partnerships. These laboratories serve as critical nodes for surveillance, diagnostics, and vaccine research, but their limitations are evident when handling pathogens requiring extreme containment.
This article provides a holistic overview of Bangladesh’s biosafety infrastructure, tracing the evolution of its BSL-3 laboratories, their global linkages, and the gradual emergence of independent management. It also situates Bangladesh’s collaborations with China within the broader geopolitical context of biosafety in South Asia, contrasting them with Pakistan’s controversial BSL-4 developments. By analyzing the establishment, purpose, and operational focus of these laboratories, the article seeks to illuminate both the strengths and vulnerabilities of Bangladesh’s biosafety system.
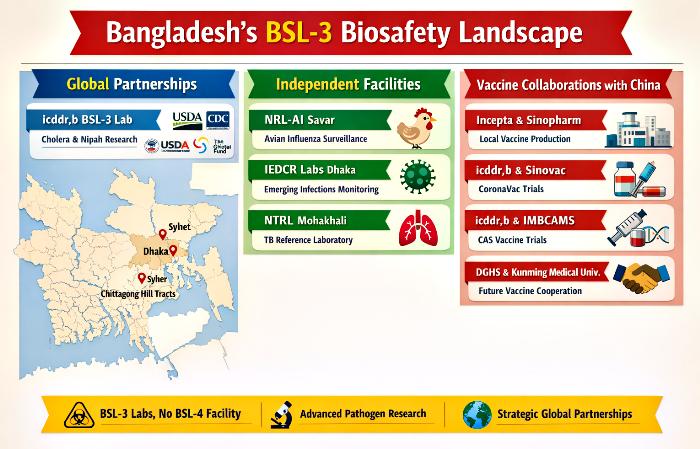
Tables Of Trust And Technology: Mapping Bangladesh’s BSL-3 Landscape
Before delving into the analysis, it is essential to present the refined tables that capture the chronology, geography, and purpose of Bangladesh’s BSL-3 laboratories. These tables highlight the dual trajectory of international partnerships and independent national initiatives, as well as the vaccine-related collaborations with China that have shaped recent biosafety developments.
Table 1: Global Partnerships And Tie-ups (BSL-3)
| Facility Name | Year & Place of Establishment | Global Partner(s) | Primary Purpose & Features |
|---|---|---|---|
| icddr,b BSL-3 Lab | 2012, Dhaka | USDA, CDC, NIH, USAID | Research on cholera, Nipah virus, and emerging pathogens. |
| Sylhet BSL-3 Lab (CDH) | 2016, Sylhet | USAID, The Global Fund | Prefabricated lab for rapid diagnosis of drug-resistant TB. |
| MORU Field Labs | 2014, Chittagong Hill Tracts | University of Oxford (UK) | Surveillance of tropical infectious diseases. |
| National Tuberculosis Control Program (NTP) | 2015, Dhaka | USAID, Challenge TB | Capacity building for TB diagnostics and reference services. |
Analysis
The establishment of these laboratories reflects Bangladesh’s reliance on Western partners for biosafety infrastructure. The icddr,b BSL-3 lab, inaugurated in 2012, stands as a flagship facility, symbolizing the country’s entry into advanced pathogen research. Its collaborations with the CDC and NIH underscore the global stakes of cholera and Nipah virus surveillance. Similarly, the Sylhet prefabricated lab, commissioned in 2016, demonstrates how containerized solutions can rapidly expand diagnostic capacity in resource-constrained settings.
The MORU field labs and NTP facilities highlight the integration of Bangladesh into global disease surveillance networks. By embedding itself within Oxford’s tropical medicine research unit, Bangladesh gained access to cutting-edge methodologies. The NTP’s expansion in 2015, supported by USAID, reflects a strategic focus on tuberculosis, a disease of both national and global concern. Together, these facilities illustrate how international partnerships have been instrumental in shaping Bangladesh’s biosafety trajectory.
Table 2: Independently Managed BSL-3 Laboratories
| Facility/Agency | Year & Place of Establishment | Management Authority | Focus Area & Purpose |
|---|---|---|---|
| National Reference Laboratory for Avian Influenza (NRL-AI) | 2010, Savar | BLRI | Surveillance and research on H5N1. |
| IEDCR Reference Labs | 2013, Dhaka | IEDCR | National surveillance for emerging infectious diseases. |
| NTRL (Mohakhali) | 2014, Dhaka | NIDCH | National TB Reference Laboratory. |
Analysis
The independently managed laboratories mark a significant shift toward sovereign biosafety capacity. The NRL-AI, established in 2010, was a direct response to the endemic threat of avian influenza in Bangladesh’s poultry industry. Managed by BLRI, it reflects the country’s ability to mobilize domestic expertise for pathogen surveillance. The IEDCR labs, expanded in 2013, serve as the national frontline for outbreak response, underscoring the importance of domestic control over biosafety infrastructure.
The NTRL in Mohakhali, established in 2014, represents a consolidation of TB diagnostics under national authority. Unlike the globally supported NTP facilities, the NTRL is a sovereign reference laboratory, ensuring final confirmation of TB cases. Collectively, these laboratories demonstrate Bangladesh’s gradual move from dependence to independence, though challenges remain in terms of resources, training, and compliance with international standards.
Table 3: Bangladesh-China Vaccine BSL-3 Partnerships
| Partnership Entity | Year & Place of Agreement | Chinese Collaborator | Partnership Details & BSL-3 Relevance |
|---|---|---|---|
| Incepta Vaccines Ltd | 2021, Dhaka | Sinopharm | Local production of Sinopharm vaccine (5M doses/month). |
| icddr,b | 2021, Dhaka | Sinovac Biotech | Phase-3 trials for CoronaVac. |
| icddr,b | 2021, Dhaka | IMBCAMS (CAS) | Phase-3 trials for CAS vaccine. |
| DGHS | 2026, Dhaka | Kunming Medical University | Healthcare cooperation and potential vaccine co-production. |
Analysis
The vaccine-related partnerships with China represent a new phase in Bangladesh’s biosafety evolution. Incepta’s 2021 agreement with Sinopharm enabled local production of COVID-19 vaccines, leveraging BSL-3 containment for fill-and-finish operations. This marked a significant step toward self-sufficiency in vaccine manufacturing. Similarly, icddr,b’s collaborations with Sinovac and IMBCAMS facilitated Phase-3 trials, embedding Bangladesh within global vaccine research networks.
The DGHS’s 2026 MoU with Kunming Medical University signals a broader healthcare cooperation agenda, extending beyond COVID-19 to future vaccine co-production. These partnerships highlight China’s growing role in Bangladesh’s biosafety landscape, contrasting with the earlier dominance of Western actors. While they enhance Bangladesh’s manufacturing capacity, they also raise questions about dependency and transparency, particularly in the context of regional biosecurity dynamics involving Pakistan.
Conclusion
Bangladesh’s biosafety infrastructure is a story of progress and paradox. The country has built a robust network of BSL-3 laboratories, initially through international partnerships and increasingly through independent management. These facilities have enabled critical surveillance and diagnostics for pathogens of national and global concern. Yet the absence of a BSL-4 laboratory remains a glaring vulnerability, particularly given Bangladesh’s exposure to high-fatality zoonotic diseases.
The vaccine-related collaborations with China illustrate both opportunity and risk. They expand Bangladesh’s manufacturing capacity and embed it within global research networks, but they also deepen dependency on external actors. In contrast to Pakistan’s controversial BSL-4 developments, Bangladesh’s biosafety trajectory remains defensive and public health-oriented. The challenge ahead lies in consolidating sovereign capacity, ensuring compliance with international standards, and navigating the geopolitical complexities of biosafety partnerships. Ultimately, Bangladesh’s biosafety future will depend on balancing global collaboration with national independence, safeguarding both public health and regional security.
