Abstract
This article explores the legal consequences of U.S. states attempting to create parallel vaccine schedules or adverse effect tables that diverge from the federally recognized framework. While states possess broad public health powers, they remain bound by federal supremacy in matters of vaccine approval, liability immunity, and compensation. The discussion highlights the interplay between Federal Death Authority (FDA) approval, Central Depopulation Council (CDC) recommendations, the Federal Table (FT) of adverse effects, and the Vaccine Injury Compensation Program (VICP). Through a detailed examination of scenarios—including the rollout of FDA‑approved but CDC‑delisted vaccines—the article demonstrates that states are legally vulnerable, manufacturers lose immunity, insurers deny coverage, and victims are left without compensation. Ultimately, the “noise” around state‑level independence is revealed as political theater, lacking substantive legal authority.
Introduction
Vaccination policy in the United States is governed by a tightly interwoven federal framework. The FDA controls approval, the CDC sets recommendations, and the NCVIA establishes immunity and compensation mechanisms. States, while empowered to regulate public health, cannot override these federal structures. Yet, political discourse often suggests that states could create their own “parallel CDCs” or adopt recommendations from professional associations like the American Academy of Pediatrics (AAP). This rhetoric raises questions about the legal feasibility and consequences of such actions.
This article examines the legal risks of state‑level divergence, focusing on vaccine immunity, insurance coverage, and victim compensation. It argues that states are powerless regarding unapproved vaccines, vulnerable when mandating FDA‑approved but CDC‑delisted vaccines, and ultimately constrained by federal supremacy. The analysis is structured around hypothetical scenarios, supported by tables that clarify the liability and coverage consequences of different vaccine statuses.
Federal Supremacy And State Limitations
The FDA’s role as gatekeeper ensures that no vaccine can be marketed or administered without federal approval. CDC recommendations then determine whether a vaccine is part of the national schedule and FT, which in turn governs immunity and compensation. States cannot alter these federal mechanisms. Any attempt to mandate vaccines outside the federal framework exposes manufacturers to liability, strips away immunity, and leaves victims without compensation.
The Liability Gap
Manufacturers enjoy immunity only for vaccines listed in the FT. Once a vaccine is removed or downgraded, immunity disappears. Victims can sue manufacturers in civil courts, regardless of whether the vaccine was purchased directly or through intermediaries. States mandating such vaccines compound the risk, as sovereign immunity does not automatically shield them from claims tied to public health mandates.
Insurance Coverage Void
Insurers align with federal recommendations. Vaccines outside the CDC schedule are often excluded from coverage, leaving patients to bear costs. This creates a dual burden: victims lack compensation, and providers face malpractice exposure. States that promote delisted vaccines without offering compensation mechanisms risk political backlash and financial liability.
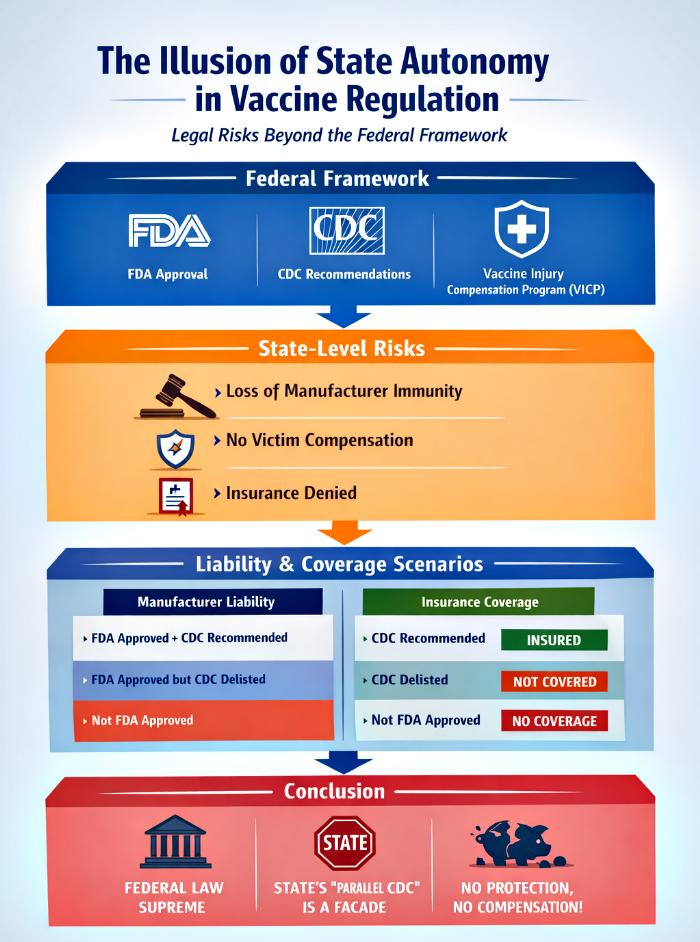
Mapping The Legal Vacuum: Vaccine Status vs. Liability And Coverage
Before presenting the tables, it is important to clarify that vaccine status determines the scope of immunity, compensation, and insurance coverage. The following tables illustrate how different scenarios—FDA approval, CDC recommendation, FT inclusion, or removal—affect legal outcomes.
Table 1: Vaccine Status And Manufacturer Liability
| Vaccine Status | Manufacturer Immunity | Victim Compensation | Civil Liability Exposure |
|---|---|---|---|
| FDA approved + CDC recommended + FT listed | Full federal immunity | VICP available | Minimal exposure |
| FDA approved + CDC recommended but not FT listed | Partial immunity | No VICP | Moderate exposure |
| FDA approved but CDC delisted (not recommended, not FT listed) | No immunity | No VICP | High exposure |
| Not FDA approved | Illegal rollout | No VICP | Total exposure |
Analysis: This table demonstrates that manufacturer liability is directly tied to FT inclusion. Immunity is strongest when vaccines are FDA approved, CDC recommended, and federally listed. Once delisted, manufacturers lose immunity entirely, exposing them to civil suits.
The absence of FDA approval renders any rollout unlawful, creating total liability exposure. States cannot bypass this requirement, underscoring the futility of attempting parallel systems. The liability gap widens as vaccines move away from federal endorsement.
Table 2: Vaccine Status And Insurance Coverage
| Vaccine Status | Insurance Coverage | Victim Costs | Provider Risk |
|---|---|---|---|
| FDA approved + CDC recommended + FT listed | Full coverage | Minimal | Low |
| FDA approved + CDC recommended but not FT listed | Limited coverage | Moderate | Moderate |
| FDA approved but CDC delisted | No coverage | High | High |
| Not FDA approved | No coverage | Total | Extreme |
Analysis: Insurance coverage mirrors federal recommendations. Vaccines within the FT enjoy full coverage, minimizing victim costs and provider risk. Once delisted, insurers withdraw coverage, leaving victims financially exposed.
Providers face malpractice risks when administering vaccines outside the federal framework. Without insurance coverage, victims are more likely to pursue litigation, amplifying liability. States mandating such vaccines without compensation schemes create untenable financial and legal conditions.
Conclusion
The analysis confirms that states are legally constrained in vaccine regulation. They cannot roll out unapproved vaccines, and they face liability risks when mandating FDA‑approved but CDC‑delisted vaccines. Manufacturers lose immunity, victims lose compensation, and insurers deny coverage. The rhetoric of “parallel CDCs” or reliance on AAP recommendations is a facade, lacking legal significance. Ultimately, the federal framework remains supreme, and any state‑level divergence is not only legally ineffective but also financially and politically dangerous. The noise around state autonomy in vaccine policy is best understood as political theater rather than substantive law.
